New Publication: Navigating HIV Treatment Failure in the Dolutegravir Era
As access to dolutegravir-based antiretroviral therapy (ART) continues to expand globally, it has transformed HIV treatment offering a potent, durable, and widely adopted regimen for millions of people. Yet as its use scales, a critical question is emerging for clinicians and policymakers alike: how should virologic failure be managed in patients receiving this first-line therapy?
A recent global review of national HIV treatment guidelines across the 50 countries with the highest adult HIV prevalence highlights a complex and evolving policy landscape. While dolutegravir-based regimens remain highly effective, instances of virologic failure though relatively uncommon are prompting divergent strategies in clinical management. These differences reflect a balance between scientific evidence, resource availability, and public health priorities, underscoring the need for clearer data to guide future decisions.
Publication Summary
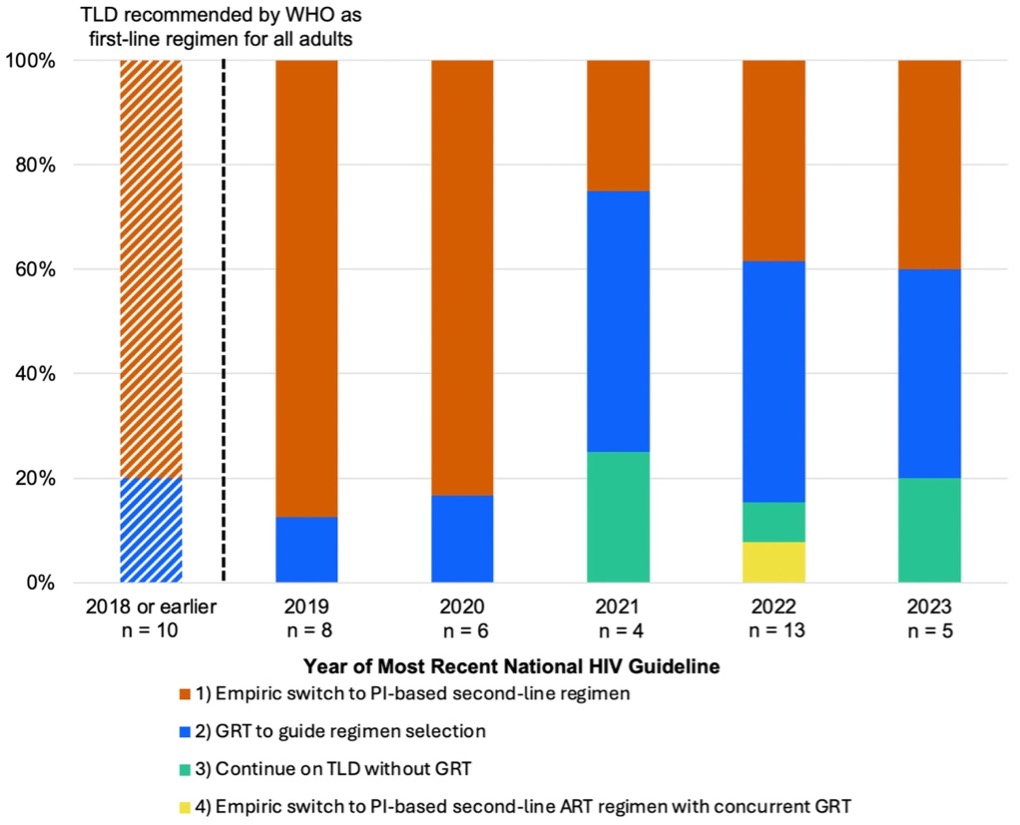
Expanded access to dolutegravir as part of a fixed-dose combination with tenofovir disoproxil fumarate and lamivudine (TLD) has revolutionized global HIV treatment, with more than 25 million people prescribed this regimen in 2024. With a team led by our clinical collaborators at Massachusetts General Hospital and Harvard Medical School, together with African and international partners (including WHO representatives), we reviewed national clinical guidelines for management of virologic failure on first-line TLD in the 50 countries with the highest prevalence of HIV among adults; recent guidelines were not available online for four. Among the remaining 46 country guidelines, we identified four distinct approaches: 1) empiric switch to a protease inhibitor (PI)-based regimen without genotypic resistance testing (GRT) (n=28; 61%); 2) GRT to guide antiretroviral therapy selection (n=14; 30%); 3) continuation of TLD without GRT (n=3; 7%); and 4) empiric switch to a PI-based regimen with concurrent GRT (n=1; 2%). The diversity of guideline-endorsed approaches underscores the critical need for additional data to inform policy regarding this important clinical scenario.
Why This Matters
For countries carrying the highest burden of HIV including many in Africa these differences in clinical guidance are not theoretical. They directly shape how millions of people are treated when first-line therapy no longer suppresses the virus.
At the heart of the issue is a fundamental trade-off. Switching patients too quickly to second-line regimens may introduce higher costs, greater side effects, and more complex treatment pathways. But delaying a switch particularly without access to resistance testing risks keeping patients on failing regimens, with implications for both individual health outcomes and the potential spread of drug-resistant virus.
The variability in national approaches also reflects deeper system realities: unequal access to diagnostic tools like genotypic resistance testing, differences in healthcare infrastructure, and the financial pressures facing HIV programmes globally. As funding constraints tighten, these decisions become even more consequential.
What this review makes clear is that the science is still catching up with policy. As new evidence emerges from ongoing clinical trials such as our own RESOLVE study and the Ndovu study it will be critical to align guidelines with what works best in practice. Doing so will help protect the long-term effectiveness of dolutegravir-based treatment, while ensuring that patients receive the most appropriate care at the right time.
To read the publication and access a full list of references, please visit: https://pmc.ncbi.nlm.nih.gov/articles/PMC12380185/
DR RICHARD LESSELS

Dr Lessells is an Infectious Diseases clinician scientist at the KwaZulu-Natal Research Innovation & Sequencing Platform (KRISP), a research centre at the University of KwaZulu-Natal in Durban, South Africa; and an honorary research associate at the Centre for the AIDS Programme of Research in South Africa (CAPRISA).
He did his clinical specialist training in Infectious Diseases and Internal Medicine in the U.K., and his PhD at the London School of Hygiene and Tropical Medicine. Dr Lessels has been actively involved in HIV and TB research in South Africa since 2007, initially in rural KwaZulu-Natal and then at UKZN since 2017. His current research includes an NIH-funded multi-country observational study of dolutegravir drug resistance in people with HIV (DTG Resist); an NIH-funded randomised controlled trial of strategies for the management of people with HIV who have virological failure on dolutegravir-based ART (RESOLVE); and an international multicentre randomised controlled trial of high-dose rifampicin for TB meningitis (HARVEST).
News date: 2026-04-09
Links:
KRISP has been created by the coordinated effort of the University of KwaZulu-Natal (UKZN), the Technology Innovation Agency (TIA) and the South African Medical Research Countil (SAMRC).
Location: K-RITH Tower Building
Nelson R Mandela School of Medicine, UKZN
719 Umbilo Road, Durban, South Africa.
Director: Prof. Tulio de Oliveira




